What Is Chlorine?
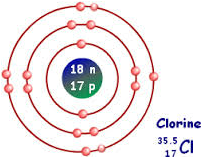 This is a Bohr-Rutherford diagram of a chlorine atom. It has 18 neutrons and 17 protons with 7 electrons in its valence shell.
This is a Bohr-Rutherford diagram of a chlorine atom. It has 18 neutrons and 17 protons with 7 electrons in its valence shell.
Chlorine (Cl) is element number 17. It is located in family 17 and period 3 on the periodic table. It has an atomic mass of 35.453 Since chlorine is located in the 3rd period that indicates it has three energy levels. Chlorine has 7 electrons in the last valence shell so it is reactive. When becoming an ion, chlorine wants to steal one electron to become an anion with a negative charge of one. Chlorine is a non-metalloid so it tends to lose electrons to metals when bonding. It is mostly found in nature bonded with sodium to form salt, usually located in saltwater.
Chlorine was discovered in 1774 by Carl Scheele but was only confirmed to be an element in 1810. Humphry Davy was the one to confirm it was an element and the one who named it. In greek, chloros means pale
green so chlorine as named after it's colour.
Chlorine was discovered in 1774 by Carl Scheele but was only confirmed to be an element in 1810. Humphry Davy was the one to confirm it was an element and the one who named it. In greek, chloros means pale
green so chlorine as named after it's colour.
Physical and Chemical Properties:
Chlorine is a greenish yellow gas at room temperature. Chlorine has a density of 3.2 g/L at 0°C.It has a boiling point at -34°C and a melting point at -101.5°C. Chlorine has a choking and irritating smell. It is also soluble in water. Chlorine has a high electronegativity and electron affinity. When it reacts with hydrogen it produces light.



